Dr. Rachel Tustin has a PhD in Education focusing on Educational Technology, a Masters in English, and a BS in Marine Science. She has taught in K-12 for more than 15 years, and higher education for ten years.
Conservation of Mass Activities
Table of Contents
- Teaching Conservation of Mass
- Inflatable Conservation of Energy
- Where Did All the Matter Go?
- The Mystery of the Inverted Balloon
Teaching students the law of conservation of mass or matter can be a challenging topic. For students, first of all, it is not always obvious that matter doesn't simply go away. And as teachers, we often don't have the resources for expensive equipment to prove to our students the law of conservation is true. So it can take some ingenuity to help students gather evidence so they can prove to themselves that mass is truly conserved.
In science classrooms, there always seems to be a few basic ingredients hanging around the storage cupboards like vinegar and baking soda. With these two ingredients, you can do all kinds of experiments, including proving to students that the law of conservation of matter does apply to the universe.
Materials
- triple beam balance
- baking soda
- vinegar
- balloons
- 16-ounce clean water bottle
- measuring cup
- funnel
Procedures
- Explain to students that the law of conservation of matter states that matter cannot be created or destroyed in a chemical reaction.
- Have students measure the mass of the empty measuring cup on the triple beam balance. Have them also measure the mass of the balloon, and their bottle. All of these measurements should be recorded in their data table.
- Next, students should add ½ cup of baking soda using the measuring cup and place it back on the triple beam balance. The difference between the mass of the cup and the mass of the cup plus baking soda will give them the actual mass of the baking soda itself.
- Add the baking soda to the bottle using a funnel. Remind students it is important that all of the baking soda gets inside the bottle.
 |
- Repeat the same process for ½ cup of vinegar.
- Students should quickly add the vinegar to the bottle and secure the balloon over the bottle opening. Students should work in pairs for this and as one student prepares to pour in the vinegar, another expands the opening of the balloon and readies to snap it over the top of the bottom as soon as the vinegar is poured into the bottle.
- As the reaction between the vinegar and baking soda happens, the balloon will fill up with gas and inflate.
- Once the reaction is done, have students measure the mass of the entire bottle and balloon filled with gas. They can record the mass in their data table.
- Have students compare the mass of their final apparatus to the mass of all of its parts.
- As a class, discuss whether or not the law of conservation of mass is true or not. Discuss how the matter in the vinegar and baking soda were transformed into gas in the balloon. The presence of the gas helps prove the law of conservation of matter to be true.
For students, there is often a myth that if you cannot see it, then it is not matter. Often when matter changes form, it is no longer obvious to the naked eye. For example, when you burn wood part it becomes carbon dioxide gas. So while it may look like matter has 'disappeared,' it, in fact, has not. Sometimes we have to force students to practice reasoning out how the law of conservation of mass is always true to help cement their understanding.
Materials
- poster board
- markers
- images that show a chemical change such as a bonfire, Mentos bubbling up soda, or a rusting piece of iron.
- pencils
- post-it notes
Procedures
- Divide your students into small groups of 3-4 students.
- Give each team a different picture from your set.
- Tell each student that whether it is obvious to the naked eye or not, matter is being conserved in each of the pictures. Review with your students the law of conservation of energy.
- Have each team discuss and decide how the chemical change in their picture supports the law of conservation of matter. Using their poster board, have each team create a flowchart to show where the matter is starting in their picture, and where it is going.
 |
- Post the final posters around your classroom. Do a gallery walk and allow other students to use the post-it notes to offer critiques of the reasoning presented.
- As an entire class, go through each poster. Discuss how the chart supports the conservation of mass. If there are any flaws in the students' reasoning, address them to benefit the entire class.
Materials
- steel wool
- vinegar
- glass flask
- balloon
- electronic scale
Procedures
- Explain to students that sometimes matter changes in ways that we can't easily observe with our eyes. Therefore, sometimes we have to come up with creative means to see how matter is changing and still being conserved.
- Have students take a piece of steel wool (about the size of a lemon) and place it in a flask. Cover the steel wool with vinegar. Explain to students that the vinegar is there to remove the coating on the steel wool so it can react with the air.
- Quickly put a balloon over the opening.
- Place the flask on a digital scale immediately and allow it to remain there while the reaction occurs. Every three minutes, have students record the mass from the scale in their data table.
- Have students continue to record the mass every three minutes until the balloon has been mostly pulled inside the flask.
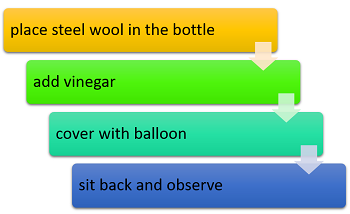 |
- Discuss with students what is happening in the experiment. For example, ask them why the balloon gets pulled inside the bottle. This phenomenon occurs because the steel wool reacts with the oxygen in the bottle, consuming it and turning it into other compounds. As the oxygen gas is converted to other types of matter, the balloon gets pulled inside the bottle due to the vacuum.
Register to view this lesson
Unlock Your Education
Become a Study.com member and start learning now.
Become a MemberAlready a member? Log In
BackResources created by teachers for teachers
I would definitely recommend Study.com to my colleagues. It’s like a teacher waved a magic wand and did the work for me. I feel like it’s a lifeline.




